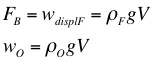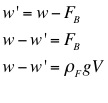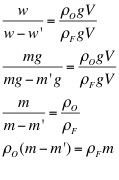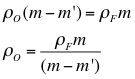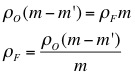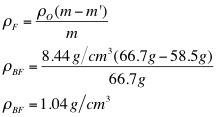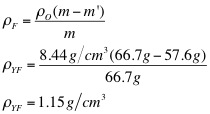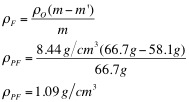Fluids
Partner: n/a
Date: 5/22/14
Date: 5/22/14
Purpose:
The purpose of this lab is to use the density, mass, and apparent mass of an object to determine the density of an unknown liquid.
The purpose of this lab is to use the density, mass, and apparent mass of an object to determine the density of an unknown liquid.
Theory:
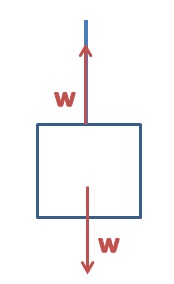
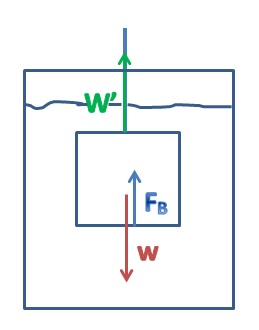
Archimedes' Principle states that the buoyant force is equal to the weight of the water that is displaced when an object is submerged. The weight of the object in the air, compared to the apparent weight of the object when it is submerged in water, can be used to determine the density of the object. If the density of the object is already known then the weight and apparent weight can be used along with the object's density to determine the density of an unknown liquid.
Archimedes' Principle states that the buoyant force is equal to the weight of the water that is displaced when an object is submerged. The weight of the object in the air, compared to the apparent weight of the object when it is submerged in water, can be used to determine the density of the object. If the density of the object is already known then the weight and apparent weight can be used along with the object's density to determine the density of an unknown liquid.
Experimental Technique:
1. Set up the balance so that the mass can be hung and choose an object. Mass the object. (See image 1)
2. Hang the object in a beaker of water and mass the object again (apparent mass).
3. Use the mass of the object, the apparent mass of the object, and the density of water to calculate the density of the object.
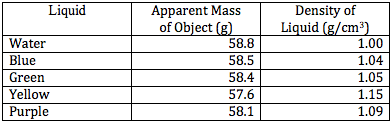
4. Hang the mass in beakers of the different liquids and record the apparent mass of the object in each liquid. (See image 2)
5. Using the density of the object, the object's mass, and the object's apparent mass, calculate the density of each liquid.
6. Change the balance so that it can be used normally.
7. Place a graduated cylinder on the balance and zero it.
8. Pour the liquid into the graduated cylinder and record the mass and volume of the liquid. (See image 3)
9. Use that mass and volume to calculate the density of the liquid.
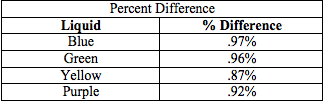
10. Compare the densities found by using the object to the densities found by using the volume by finding the percent difference.
1. Set up the balance so that the mass can be hung and choose an object. Mass the object. (See image 1)
2. Hang the object in a beaker of water and mass the object again (apparent mass).
3. Use the mass of the object, the apparent mass of the object, and the density of water to calculate the density of the object.
4. Hang the mass in beakers of the different liquids and record the apparent mass of the object in each liquid. (See image 2)
5. Using the density of the object, the object's mass, and the object's apparent mass, calculate the density of each liquid.
6. Change the balance so that it can be used normally.
7. Place a graduated cylinder on the balance and zero it.
8. Pour the liquid into the graduated cylinder and record the mass and volume of the liquid. (See image 3)
9. Use that mass and volume to calculate the density of the liquid.
10. Compare the densities found by using the object to the densities found by using the volume by finding the percent difference.
Data:
Analysis:
Density of Object
Method 1
Method 2
Percent Difference
Conclusion:
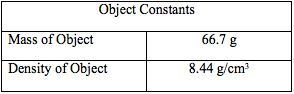
The purpose of this lab was to determine the density of the unknown fluids using Archimedes' Principle. First, the density of the object was found to be 8.44 g/cm3. Then, using the apparent mass of the object in each fluid, as well as its density, the density of each fluid was determined. The densities of each fluid are as follows: blue 1.04 g/cm3, green 1.05 g/cm3, yellow 1.15 g/cm3, and purple 1.09 g/cm3. Next, the densities were determined solely by using the mass and volume of the liquids. Using the second method, the density of the blue fluid was found to be 1.03 g/cm3. This yielded a .97% difference. The density of the green fluid was 1.04 g/cm3, which yielded a .96% difference. The yellow fluid has a density of 1.14 g/cm3, using the second method, and had a percent difference of .87%. Finally, the purple fluid's density was found to 1.08 g/cm3, which yielded a .92% difference.
The purpose of this lab was to determine the density of the unknown fluids using Archimedes' Principle. First, the density of the object was found to be 8.44 g/cm3. Then, using the apparent mass of the object in each fluid, as well as its density, the density of each fluid was determined. The densities of each fluid are as follows: blue 1.04 g/cm3, green 1.05 g/cm3, yellow 1.15 g/cm3, and purple 1.09 g/cm3. Next, the densities were determined solely by using the mass and volume of the liquids. Using the second method, the density of the blue fluid was found to be 1.03 g/cm3. This yielded a .97% difference. The density of the green fluid was 1.04 g/cm3, which yielded a .96% difference. The yellow fluid has a density of 1.14 g/cm3, using the second method, and had a percent difference of .87%. Finally, the purple fluid's density was found to 1.08 g/cm3, which yielded a .92% difference.
References:
Buoyancy. (n.d.). Pressure. Retrieved May 26, 2014, from http://hyperphysics.phy-astr.gsu.edu/hbase/pbuoy.html
Buoyancy. (n.d.). Pressure. Retrieved May 26, 2014, from http://hyperphysics.phy-astr.gsu.edu/hbase/pbuoy.html